Welcome to the homepage of the Institute for Developmental Immunology (IDI) at the Biocenter of the Medical University of Innsbruck. The IDI is currently headed by Prof. Andreas Villunger, PhD, who was appointed as a head of this unit in 2007 as junior professor and is now full professor for Developmental Immunology at MUI.
.
The Institute is organized in four research groups, run by head Andreas Villunger and assistant/associate professors Jan Wiegers, Joel Riley, Verena Labi and Sebastian Herzog. Please click the logo to learn more about the respective groups!
With their coworkers, IDI investigators explore basic mechanisms of immune cell development and differentiation with a focus on microRNAs and steroid hormones. In addition, we are interested in studying general principles of cellular transformation, focusing on the role of BCL2-regulated cell death and the p53 signalling network as barrier against malignant disease.
All group leaders are involved in training undergraduate as well as graduate students of different life-science disciplines, including biochemistry, biology as well as molecular and human medicine. If you are interested in research training opportunities at different levels (summer students, BSc, MSc, or MD/PhD), or have any teaching related questions, please contact the respective group leader.
Recent News

We are happy to announce that Valentina Sladky was awarded with one of the prestigious ESPRIT (Early-Stage Program: Research - Innovation - Training) grants by the FWF. In the next three years, she will investigate how polyploidization, the process in which a cell gains more than two complete sets of chromosomes, is regulated across and shapes mammalian tissues. Congrats!
This year, we skipped the Alps and met with our team members from Vienna along the Danube in Maria Taferl at hotel Schachner for our annual lab retreat. Going totally green, we either travelled by train and some decided to go there by bicycle even. It was a very relaxing and stimulating event for all of us, before the summer break. A big thanks also to Stephan Geley and Michael Schubert for joining and providing critical feedback and adding to the lively discussions…….
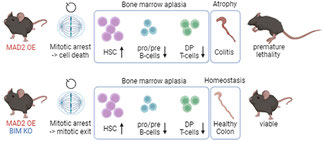
During her PhD, Gerlinde analyzed the consequences of chronic spindle assembley checkpoint activation in vivo and realued that differnt tissues are
differently affected with fast proliferating cell types in the gastrointestinal tract of bone marrow, being most affected. Looking into causes and consequences upon MAD2 overexpression, she
realized that delays in mitosis cause apoptosis that can be blocked by BCL2 overexpression or loss of the BCL2 family protein BIM, in a rather tissue dependent manner. This work has been
published earlier this year in EMBO reports. Read more...
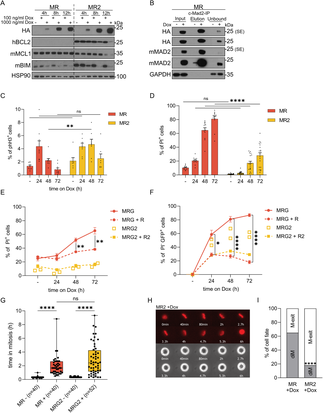
Work initiated by a former post doc in our lab, Fabian Schuler, was brought to completion in a concerted effort by Vincent and Gerlinde, now published in Science
Advances. In their work, they could show that extra centrosomes, frequently seen in human cancer can become toxic to primary cells and promote cell death. This phenomenon in part explains the
fact that extra centrosomes are selected against in murine models of blood cancer, driven by oncogenes such as MYC or vABL. Moreover, Vincent realized that in models of DNA damage driven cancer
centrosome amplification even delayed cancer, constrasting current believe that these structures are primarily pro-tumorigenic. Here, the caspase-2 PIDDosome was able to sensitize lympoohatic
cells to mitochondrial apoptosis, most pronounced when combined with DNA damage, leading to the inducion of rapid cell death and delayed tumor onset in a model of irradiation-driven blood cancer.
Read more…..

Centrioles are frequently amplified in early B cell development but dispensable for humoral immunity
We are happy to report that Marina´s PhD work on the dynamic regulation of centriole numbers in developing B cells has now
been published. Using different genetic models she noted that early progenitor B cells accumulate centrosomes that are then cleared and no longer detectable
in mature B cells. Surprisingly, loss of centrosomes abrogates B cell development but development can be restored by blocking p53-dependent apoptosis in progenitor B cells. These B cells are
competent to produce antibodies in response to model antigens, suggesting that B cells in germinal centers that divide rapidely do not rely on the formation of a mitotic spindle organized by
centrosomes. Fantastic collaboration with the lab of Andrew Holland, formerly at Johns Hopkins Medical School and the Labi group at our insitute. Read more...